The inherent heterogeneity of diseases such as cancer is driving the field of personalized (precision) therapies. Within personalized therapies, chimeric antigen receptor (CAR-T) therapies are gaining traction. With the approval of Novartis’ Kymriah, Singapore is now all set to deliver the transformative treatment for cancers and takes the lead in medical tourism within Asia-Pacific (APAC), says GlobalData, a leading data, and analytics company.
In March 2021, Novartis received approval for Kymriah for adult r/r diffuse large B-cell lymphoma DLBCL (DLBCL) patients and young adult r/r B-cell acute lymphoblastic leukemia (ALL) patients. Kymriah will be the first CAR-T therapy to be approved in Southeast Asia.

Novartis, Gilead, and BMS are the top CAR-T therapy companies in APAC. Kymriah was approved in Australia in December 2018, and in Japan in March 2019.
Kymriah approval in Singapore is the third approval of a CAR-T therapy this year in APAC. In January, Gilead’s Yescarta was approved for DLBCL in Japan, In March, BMS’ Breyanzi was approved for relapsed/refractory (R/R) large B-cell lymphomas (LBCL) and R/R follicular lymphoma in Japan.
Bhavani Nelavelly, Pharma Analyst at GlobalData, comments: “CAR-T therapies are evolving as disruptors in the next generation of cancer treatment. With the approval of Kymriah, Singapore has made history in Southeast Asia with the first commercial approval of CAR-T therapy, which is a significant milestone in precision oncology, offering an effective therapy option for relapsed/refractory DLBCL and ALL patients. Kymriah is a complex personalized therapy that needs collaboration between several stakeholders. However, the medical infrastructure in Singapore ensures effective drug delivery within treatment centers.”
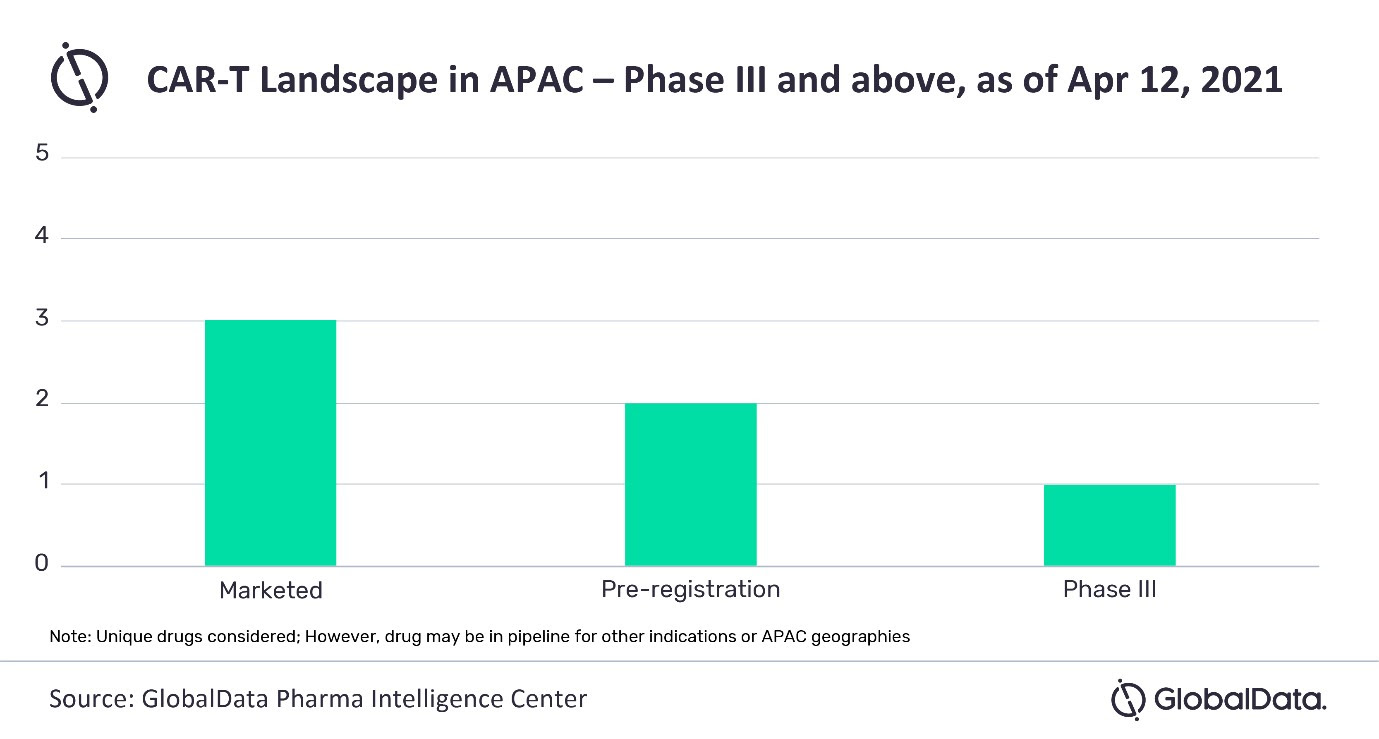
According to GlobalData’s Pharma Intelligence Center, there are six CAR-T therapies in Phase III stage and above in APAC.
Singapore is a popular medical tourism destination within APAC. Other popular countries are Thailand, Malaysia, India, and South Korea.
Nelavelly continues: “The approval of CAR-T therapy in Singapore further makes it first among the popular medical tourism destinations within APAC.”
Currently, the treatment is available in Singapore General Hospital for adult r/r DLBCL and young adult r/r B-cell ALL, while discussions with the National University Hospital are underway to expand the availability of Kymriah for both adult and pediatric patients
Nelavelly concludes: “The approval of Kymriah will bring new hope to the patients, whose cancer advanced after the previous treatment. However, with a high price (the price of Kymriah in Japan is US$306,000) and logistics, and less population eligible to receive the treatment, CAR-T therapies will require financial assistance to make them affordable.”






